
Luteinizing hormone (LH) suppression to prepubertal levels1
Percentage of Patients Achieving Prepubertal LHa
(GnRH-Stimulated LH ≤5 IU/L)1,b
-
Month 1
-
Month 6
(primary endpoint) -
Month 12
93% of children achieved LH
suppression with TRIPTODUR at
Month 6 (primary endpoint)1
TRIPTODUR demonstrated
LH suppression to prepubertal
levels as early as Month 11
98% of children achieved LH suppression with TRIPTODUR at Month 121
Three patients presented with nonsuppressed LH levels at Month 6.2
- Two of these nonresponders showed prepubertal levels at month 12, one of whom had a borderline LH value of 5.1 IU/L at month 6 but a suppressed testosterone level of 2 ng/dL. The other encountered a technical problem with the first injection, which is likely to have played a significant role regarding this treatment failure.
- The third nonresponder was a 9-year-old overweight boy with a BMI of 23.1 kg/m2, who may have required a higher drug dose for adequate hormonal suppression, or may represent one of the rare children with CPP who do not achieve suppression with GnRH-agonists.
Note: 95% of children (n/N=42/44) achieved prepubertal LH levelsa at Months 2, 3, and 9.
a Serum LH ≤5 IU/L thirty minutes after GnRH-agonist stimulation.
b Results are from intent-to-treat (ITT) population.
Quick Links
Learn about dosing and administrationEffective suppression of stimulated LH2
Mean LH Levels for Girls and Boys (Intent-to-Treat Population)2
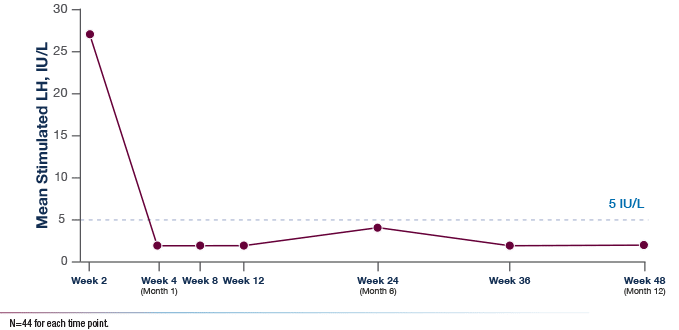
TRIPTODUR demonstrated
effective suppression of
stimulated LH throughout the study2
Mean LH remained
suppressed to prepubertal
levels (≤5 IU/L) at all time
points post-initiation of
TRIPTODUR2
Please go to section labeled “LH suppression to prepubertal levels” for more information about treatment nonresponders.
Effective suppression of sex steroids2
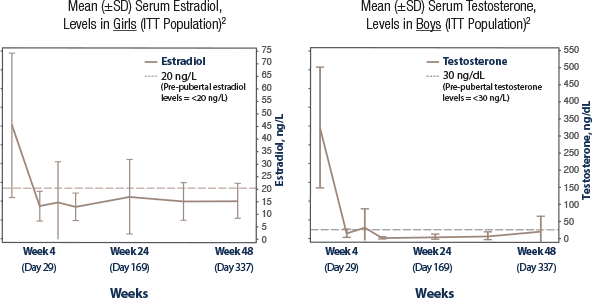
TRIPTODUR demonstrated
effective suppression
of sex steroids2
Suppression was achieved
from Week 4 through Week 482
Clinical signs of puberty

TRIPTODUR arrested or reversed
progression of clinical signs of puberty1,2
77% of girls
(n/N=30/39) exhibited regression of uterine
length at Month 121
100% of boys
(n/N=5/5) showed absence of progression
of testicular volume at Month 121
aResults are from ITT population.
bMonth 6 data: 64% (n/N=28/44).
cMonth 6 data: 91% (n/N=40/44).
*Tanner stage refers to a scale used to measure the onset and progression of
pubertal changes as determined by the physical development of secondary sex characteristics.3
Study Design
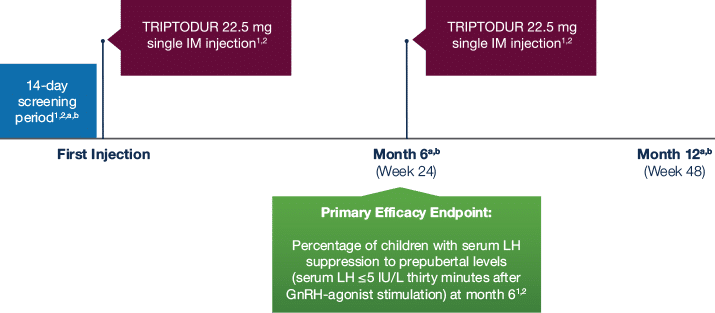
The efficacy and safety
of TRIPTODUR were
demonstrated in a multicenter
phase 3 study1,2
-
The study was an open-label, noncomparative, multicenter phase 3 trial on the efficacy and safety of triptorelin 22.5 mg 6-month formulation in patients with CPP. The study was conducted in 44 patients (n=39 girls; n=5 boys) with CPP aged 2 to 9 years who were naive to previous GnRH-agonist treatment.1,2
-
TRIPTODUR was initiated no more than 18 months after onset of the first signs of puberty.2
-
Eighteen medical centers in the United States, Chile, and Mexico participated in the study.2
Measurements Taken:
a Measurement of serum LH, FSH, estradiol (girls), and testosterone (boys) levels collected at screening and at Months 1 (Day 29), 2 (Day 57), 3 (Day 85), 6 (Day 169), 9 (Day 253), and 12 (Day 337).
1,2 LH and FSH were assessed both before and 30 minutes after stimulation with commercial leuprolide acetate 20 μg/kg subcutaneous injectable solution 1 mg/0.2 mL.
b Transabdominal ultrasound was performed in girls; bone age was assessed by x-ray of the left hand and wrist. Collected during screening period, Month 6, and Month 12.
2 Presence of unstable intracranial tumors at screening was excluded by MRI or CT scans of the brain, unless they had been performed within 3 months prior to treatment initiation.
BA, bone age; BMI, body mass index; CA, chronological age; CPP, central precocious puberty; CT, computerized tomography; FSH, follicle-stimulating hormone; GnRH, gonadotropin-releasing hormone; IM, intramuscular; ITT, intent-to-treat; LH, luteinizing hormone; MRI, magnetic resonance imaging.



